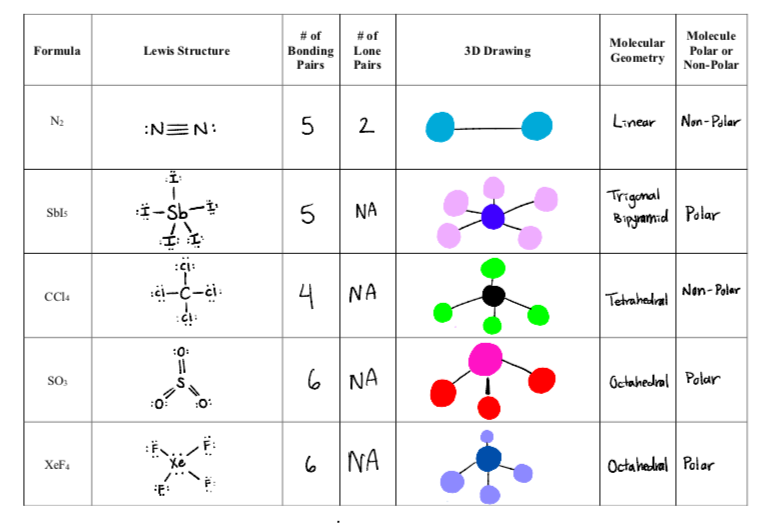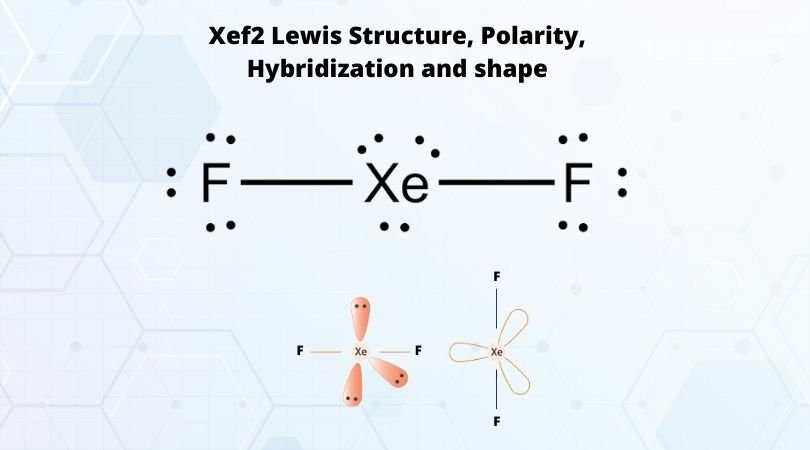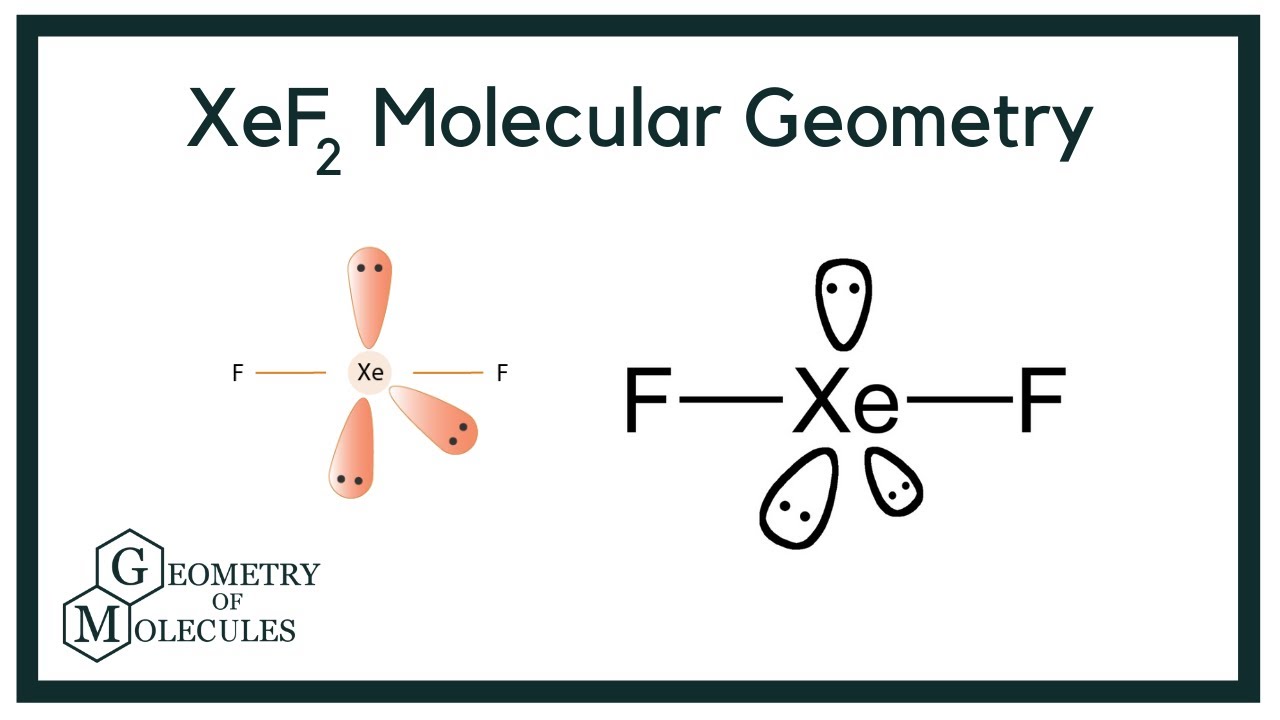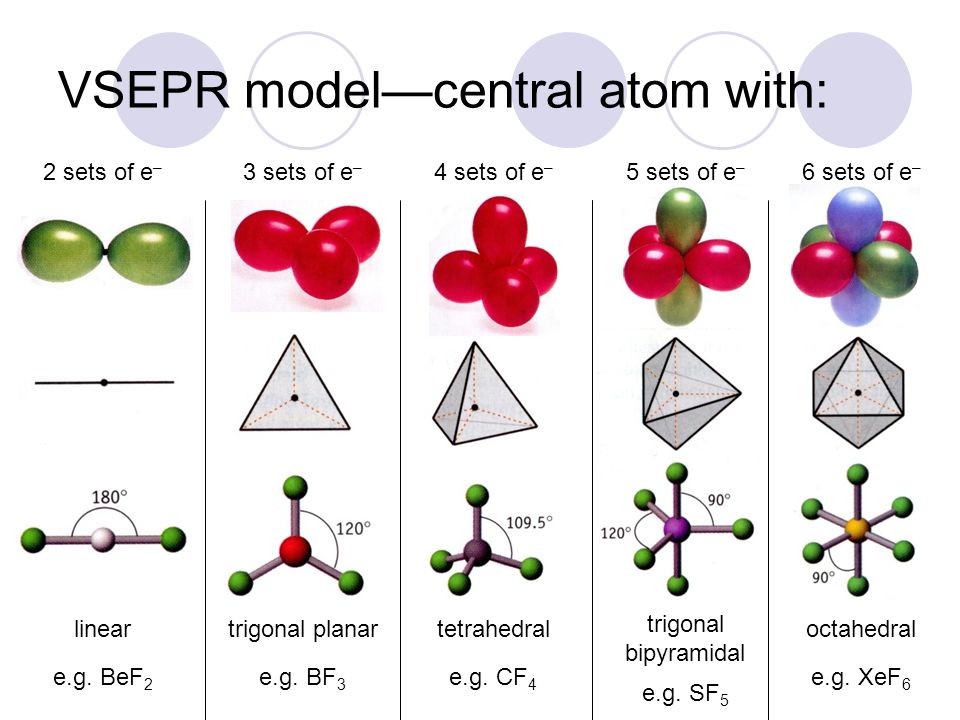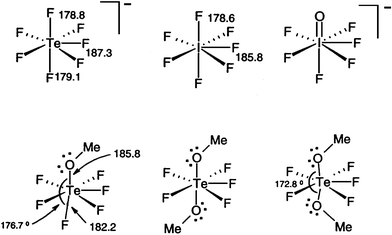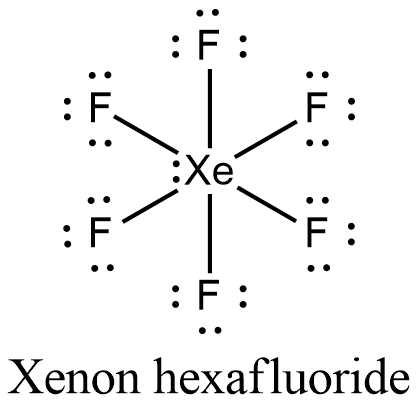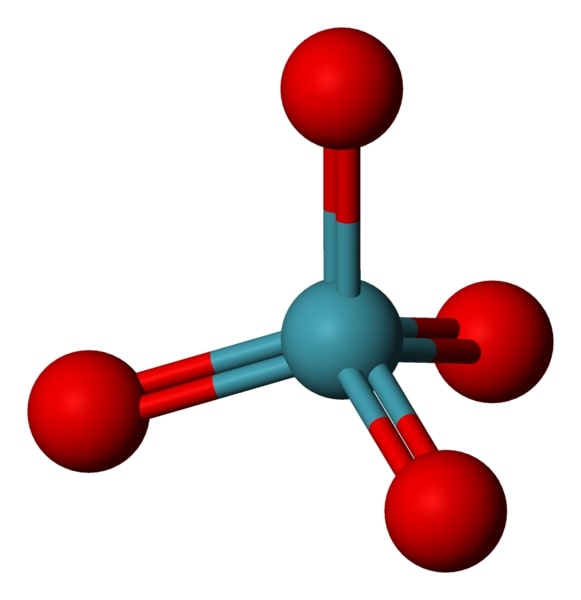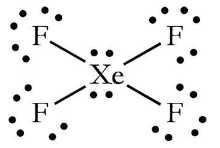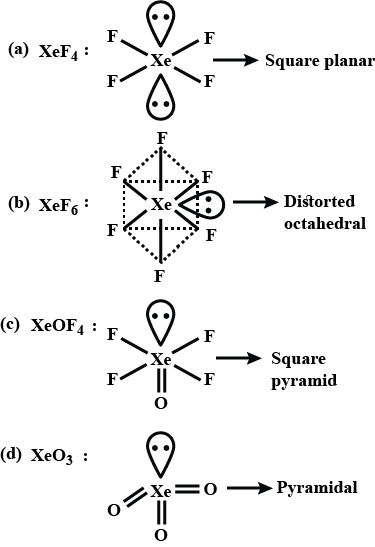
Match the Xenon compounds in Column - I with its structure in Column - II and assign the correct code:Column - IColumn - II(a) XeF4 (i)Pyramidal(b) XeF6 (ii)Square planer(c) XeOF4 (iii)Distorted octahedral(d)

XeF6 Hybridization (Xenon Hexafluoride) | XeF6 Hybridization (Xenon Hexafluoride) Were you looking for a detailed video to help you determine the hybridization of Xenon Hexafluoride? If yes,... | By Geometry Of Molecules | Facebook

![Solution] 5) Which of the following is the best Lew… | Wizeprep Solution] 5) Which of the following is the best Lew… | Wizeprep](https://d3rw207pwvlq3a.cloudfront.net/attachments/000/016/739/original/graf35.png?1536159579)